Pneumococcal polysaccharide vaccine (PPSV23) - what you need to know
All content below is taken in its entirety from the CDC Pneumococcal Polysaccharide Vaccine Information Statement (VIS): CDC review information for Pneumococcal Polysaccharide VIS: www.cdc.gov/vaccines/hcp/current-vis/pneumococcal-polysaccharide.html
Images
I Would Like to Learn About:
Information
1. Why get vaccinated?
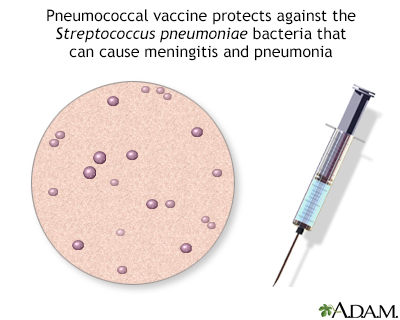
Pneumococcal polysaccharide vaccine (PPSV23) can prevent pneumococcal disease.
Pneumococcal disease refers to any illness caused by pneumococcal bacteria. These bacteria can cause many types of illnesses, including:
- Pneumonia (infection of the lungs)
- Ear infections
- Sinus infections
- Meningitis (infection of the tissue covering the brain and spinal cord)
- Bacteremia (bloodstream infection)
Anyone can get pneumococcal disease, but young children, older adults, and people with certain risk factors are at the highest risk.
Most pneumococcal infections are mild. However, some can result in long-term problems, such as brain damage or hearing loss. Meningitis, bacteremia, and pneumonia caused by pneumococcal disease can lead to death.
2. PPSV23
PPSV23 helps protect against 23 types of bacteria that cause pneumococcal disease.
PPSV23 is recommended as an option for some children and adolescents with certain medical conditions or risk factors.
For adults, PPSV23 is recommended following a dose of 15-valent pneumococcal conjugate vaccine (PCV15).
Your health care provider can give you more information.
3. Talk with your health care provider
Tell your vaccine provider if the person getting the vaccine:
- Has had an allergic reaction after a previous dose of PPSV23, or has any severe, life-threatening allergies.
In some cases, your provider may decide to postpone PPSV23 vaccination to a future visit.
People with minor illnesses, such as a cold, may be vaccinated. People who are moderately or severely ill should usually wait until they recover before getting PPSV23.
Your provider can give you more information.
4. Risks of a vaccine reaction
- Redness, swelling, pain, or tenderness where the shot is given; tiredness; headache; fever; chills; or muscle aches can happen after PPSV23.
People sometimes faint after medical procedures, including vaccination. Tell your provider if you feel dizzy or have vision changes or ringing in the ears.
As with any medicine, there is a very remote chance of a vaccine causing a severe allergic reaction, other serious injury, or death.
5. What if there is a serious problem?
An allergic reaction could occur after the vaccinated person leaves the clinic. If you see signs of a severe allergic reaction (hives, swelling of the face and throat, difficulty breathing, a fast heartbeat, dizziness, or weakness), call 9-1-1 and get the person to the nearest hospital.
For other signs that concern you, call your health care provider.
Adverse reactions should be reported to the Vaccine Adverse Event Reporting System (VAERS). Your health care provider will usually file this report, or you can do it yourself. Visit the VAERS website (vaers.hhs.gov) or call 1-800-822-7967. VAERS is only for reporting reactions, and VAERS staff do not give medical advice.
6. How can I learn more?
- Ask your health care provider.
- Call your local or state health department.
- Visit the website of the Food and Drug Administration (FDA) for package inserts and additional information.
Contact the Centers for Disease Control and Prevention (CDC):
- Call 1-800-232-4636 (1-800-CDC-INFO).
- Visit CDC's vaccine website.
References
Centers for Disease Control and Prevention website. PPSV23 Vaccine VIS. Pneumococcal Polysaccharide Vaccine (PPSV23): what you need to know. www.cdc.gov/vaccines/hcp/current-vis/pneumococcal-polysaccharide.html. Updated May 29, 2025. Accessed June 4, 2025.
BACK TO TOPReview Date: 7/15/2024
Reviewed By: Frank D. Brodkey, MD, FCCM, Associate Professor, Section of Pulmonary and Critical Care Medicine, University of Wisconsin School of Medicine and Public Health, Madison, WI. Also reviewed by David C. Dugdale, MD, Medical Director, Brenda Conaway, Editorial Director, and the A.D.A.M. Editorial team. Editorial update 06/04/2025.

Health Content Provider
06/01/2025
|
A.D.A.M., Inc. is accredited by URAC, for Health Content Provider (www.urac.org). URAC's accreditation program is an independent audit to verify that A.D.A.M. follows rigorous standards of quality and accountability. A.D.A.M. is among the first to achieve this important distinction for online health information and services. Learn more about A.D.A.M.'s editorial policy, editorial process and privacy policy. A.D.A.M. is also a founding member of Hi-Ethics. This site complied with the HONcode standard for trustworthy health information from 1995 to 2022, after which HON (Health On the Net, a not-for-profit organization that promoted transparent and reliable health information online) was discontinued. |
The information provided herein should not be used during any medical emergency or for the diagnosis or treatment of any medical condition. A licensed medical professional should be consulted for diagnosis and treatment of any and all medical conditions. Links to other sites are provided for information only -- they do not constitute endorsements of those other sites. © 1997- 2025 A.D.A.M., a business unit of Ebix, Inc. Any duplication or distribution of the information contained herein is strictly prohibited.