Menopause - InDepth
Hormone replacement therapy - InDepth; Estrogen - InDepth; Hormone replacement therapy - InDepth; Perimenopause - InDepth; Postmenopause - InDepthAn in-depth report on the treatment of menopause-related symptoms.
- Highlights
Menopause
Menopause is a natural process that occurs as a woman's ovaries stop producing eggs and the production of female hormones (estrogen and progesterone) declines. Menopause can also occur if a woman's ovaries are damaged by certain diseases or cancer treatments, or if they are surgically removed.
Natural menopause usually develops gradually between the ages of 45 to 55. During this transition time, called "perimenopause," menstrual periods become more irregular and begin to taper off. After menstrual periods have stopped for 12 months, a woman is considered to have reached menopause. On average, American women reach menopause around the age of 51, but menopause can occur at younger or older ages.
Perimenopausal Symptoms
During perimenopause, women may have various symptoms. Symptoms differ among women and may range from mild to severe. Hot flashes, an intense sudden build-up of body heat, are the most common symptom. Some women have no symptoms.
Treatment
Menopause is a natural condition. It is not a disease that needs medical treatment. However, some women seek treatment for the relief of perimenopausal symptoms.
Hormone therapy (HT) is the most effective drug treatment for hot flashes, but it is still a controversial subject. Certain HT regimens may increase the risks of some health conditions, including breast cancer, blood clots in the legs or lungs, and possibly cardiac disease and stroke. HT dose, duration, regimen, and route of administration may vary according to individual needs. Therefore, the best solution is to talk with your provider about the risks and benefits you might have from taking HT.
- Introduction
Menopause is a natural process that occurs as a woman's ovaries stop producing eggs, and the production of hormones such as estrogen and progesterone declines. Menopause can also occur if a woman's ovaries are surgically removed or if they are damaged by certain diseases or cancer treatments.
Natural menopause does not occur suddenly. A period called perimenopause usually begins a few years before the last menstrual cycle. There are two stages in the transition:
- Early Stage. Perimenopause can begin in some women in their 30s, but most often it starts in women ages 40 to 44. It is marked by changes in menstrual flow and in the length of the cycle. There may be sudden surges in estrogen.
- Late Stage. The late stages of perimenopause usually occur when a woman is in her late 40s or early 50s. In the late stages of the menopausal transition, women begin missing periods until they finally stop. About 6 months before menopause, estrogen levels drop significantly. The fall in estrogen triggers the typical symptoms of vaginal dryness and hot flashes (which can last from half a year to more than 5 years after onset of menopause).
Menopause is considered to have occurred after a woman has gone a full 12 months without a period. Menopause marks the end of menstruation and a women's fertility. American women reach menopause at an average age of 51 years, although it can occur as early as age 40 to as late as the early 60s. Women now have a life expectancy of more than 80 years. Currently, women can expect to live some 30 or 40 years of their lives in the postmenopausal state.
Menopause is not a disease. However, many conditions are associated with estrogen depletion, including heart disease, osteoporosis, and other complications. Fortunately, effective treatments are available for these conditions.
In a number of studies, many women have reported menopause as a positive experience and have welcomed it as a sign of a new stage in life.
- Causes
Causes of Natural Menopause
Menopause is a normal part of the aging process. It occurs naturally after a woman's ovaries stop producing eggs and releasing hormones necessary for fertility.
The ovaries are two small, almond-shaped organs located on either side of the uterus. They are key components of a woman's reproductive system. The ovaries contain 200,000 to 400,000 follicles, tiny sacks that contain immature eggs. Each month during the menstrual cycle, the ovaries release an egg, which travels through the fallopian tube to the uterus. As a woman ages, her supply of eggs decreases. Eventually, the ovaries stop releasing eggs and her monthly menstrual periods cease. The word menopause comes from the Greek words "mens" for month and "pausis" for cessation.
The ovaries produce two major female hormones: estrogen and progesterone. As a woman enters menopause, the ovarian production of these hormones declines and fluctuates until it eventually stops.
Estrogen
Estrogen has different forms. The strongest form is estradiol. Other important, but less powerful estrogens are estrone and estriol. Most of the estrogens in the body are produced by the ovaries, but they can also be formed by other tissues, such as body fat, skin, and muscle. Estrogens are essential for the reproductive process. They also affect and help regulate function in other parts of the body including:
- The brain and central nervous system
- The bones
- The liver
- The urinary tract
Progesterone
Progesterone, the other major female hormone, is necessary for preparing the uterine lining for the fertilized egg.
Hormonal Changes in Menopause
Menopause is marked by the following hormonal changes:
- Ovarian secretion of estrogen and progesterone ends.
- Although the ovaries stop producing estrogen, they still continue to produce small amounts of the male hormone testosterone, which can be converted to estrogen (estradiol) by an enzyme found in body fat.
- The adrenal gland continues to produce androstenedione (a male hormone), which is converted to estrone and estradiol in the body fat.
- The total estrogen produced after menopause is, however, far less than that produced during a woman's reproductive years.
Causes of Premature Menopause
Premature (early) menopause is menopause that occurs before age 40. Women who have a family history of premature menopause are genetically more susceptible to starting menopause early. Premature menopause can also result from:
- Surgical Removal of the Uterus, Ovaries, or Both. A hysterectomy (surgical removal of the uterus) that also involves removal of both ovaries (bilateral oophorectomy) causes immediate menopause. Women who have a hysterectomy but keep one or both ovaries do experience immediate menopause in the sense that their menstrual cycles stop. But until their ovaries fail, generally close to the time this normally occurs, they will not have other common symptoms associated with menopause caused by a lack of estrogen.
- Cancer Treatments. Some types of cancer radiation and chemotherapy treatments can induce menopause.
- Medical Conditions. Certain autoimmune disorders, such as rheumatoid arthritis and Graves disease, can cause premature menopause. Genetic disorders such as Turner syndrome that cause ovarian abnormalities can also predispose to early menopause.
- Symptoms
The most prominent symptoms of the transition to menopause include:
- Irregular menstrual periods. In the years preceding menopause, women experience changes in their menstrual cycle. The time between periods can become shorter or longer and periods may last a longer or shorter number of days. Bleeding can be heavier or lighter and change in flow from month to month. It is also normal for periods to skip a cycle. (Note: bleeding after menopause is not normal and may be a sign of a serious condition.)
- Hot flashes and night sweats. Women often feel hot flashes as an intense build-up in body heat, followed by sweating and chills. The medical term for these sensations is vasomotor symptoms. Some women report accompanying anxiety as the sensation builds. In most cases, hot flashes continue to occur for 3 to 5 years, although they may linger in some women for years after menopause. Women who have surgical removal of both ovaries, and who do not receive hormone therapy, may have more severe hot flashes than women who enter menopause naturally.
- Difficulty sleeping. Insomnia is common during perimenopause. It may be caused by the hot flashes, or it may be an independent symptom of hormonal changes.
- Vaginal dryness. Decrease in estrogen can cause thinning and drying of the vaginal membrane (vaginal atrophy also called atrophic vaginitis), which can cause itching and pain during sex (dyspareunia).
- Mood changes. Mood changes and irritability are usually due to a combination of sleeplessness and hormonal swings. Some women find the menopausal transition to be psychologically stressful; some develop clinical depression. Women may be more at risk if they have experienced severe PMS mood swings or have a history of clinical depression. Perimenopausal depression usually goes away within a few years after menopause. In general, depression is less common during the postmenopausal years than in the premenopausal ones.
Although forgetfulness is often mentioned as a symptom, evidence indicates that menopause does not affect memory or other cognitive functions. It is normal for both men and women to experience some memory lapses as part of the natural aging process.
- Complications
The decline in estrogen after menopause can increase the risk for a number of health problems for women.
Menopause and Heart Health
Coronary artery disease is the number one killer of women. Although young women have a much lower risk for coronary artery disease than young men, after menopause women catch up. After age 60, women's risk of dying from heart disease is very close to that of men. Estrogen loss is believed to play a major role in this increased risk.
Women who reach menopause before the age of 35 have a significant increase in risk for heart disease as they age. This increase is primarily due to a rise in levels of LDL ("bad") cholesterol and triglycerides, and a decrease in levels of HDL ("good") cholesterol.
Menopause and Bone Density
Osteoporosis is a disease of the skeleton in which bones become "thin" and prone to fracture. In other words, the bone loses calcium and density. At age 65, about 30% of women have osteoporosis, and nearly all of them are unaware of their condition. After age 80, up to 70% of women develop osteoporosis. Osteoporosis is a major risk factor for fracture in the spine and hip. The decrease in bone density can also lead to bone loss in the jaw and subsequent tooth decay.
Urinary Incontinence
The drop in estrogen levels brought on by menopause can thin the linings of the urethra and bladder, which may contribute to both stress and urge (overactive bladder) types of urinary incontinence.
Urinary Tract Infections
Because of low estrogen levels causing vaginal atrophy, women are at increased risk for recurrent urinary tract infections after menopause.
Weight Gain
Weight gain is common during a woman's middle-aged years, especially between the ages of 50 to 59. The hormonal changes associated with menopause contribute to these body changes. Gaining weight around the abdomen (the so-called apple shape) is a specific risk factor for heart disease, diabetes, and many other health problems. Higher body mass index (BMI) may also be associated with increased risk for more frequent or severe hot flashes.
Skin and Hair Changes
Estrogen loss can contribute to slackness and dryness in the skin and wrinkles. Many women experience thinning of their hair and some have temporary hair loss.
- Lifestyle Changes
Simple changes in lifestyle and diet can help control menopausal symptoms such as hot flashes:
- Avoid hot flash triggers like spicy foods, hot beverages, caffeine, and alcohol.
- Dress in layers so that clothes can be removed when a hot flash occurs.
- Lower room temperatures and consume plenty of cool drinks. A bedside fan can be helpful for dealing with night sweats.
- For vaginal dryness, non-estrogen lubricants and moisturizers are available without a prescription.
Heart Health Recommendations
When women reach menopause, they are at increased risk for heart disease. A heart-healthy diet and other lifestyle changes are important ways to control cholesterol and blood pressure levels.
Heart-healthy recommendations include:
- Quit smoking. If a woman smokes, she should quit. Smoking is linked to a decline in estrogen levels. Women who smoke reach menopause about 2 years earlier than nonsmokers. Smoking doubles a woman's odds of developing heart disease and is a major risk factor for osteoporosis.
- Balance calorie intake and physical activity to achieve or maintain a healthy body weight. Try to get at least 30 minutes, and preferably 60 to 90 minutes, of daily exercise. Aerobic exercise will reduce the risk of heart disease, while weight-bearing and resistance exercise will reduce the risk of osteoporosis.
- The Mediterranean diet is an excellent dietary choice for almost everyone. The main principles of the Mediterranian diet are listed below.
- Make vegetables, fruits, and whole grains the focus of your diet. Vegetables and fruits that are deeply colored (such as spinach, carrots, peaches, and berries) have the highest nutritional content. Good whole grain choices include whole wheat, oats/oatmeal, rye, barley, brown rice, buckwheat, bulgur, millet, and quinoa.
- Eat fish, especially oily fish, at least twice a week (about 8 ounces/week). Oily fish such as salmon, mackerel, and sardines are rich in the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Consumption of these fatty acids is linked to a reduced risk of sudden death and death from coronary artery disease.
- Limit daily intake of saturated fat (found in red meat, butter, cheese and whole-fat dairy products) and avoid trans fats (found in many commercially baked products, and many fast foods). Better choices are low-fat dairy products, poultry, fish, legumes (beans), and nuts. Unsaturated fats are found in plant (olive, canola) and fish oils as well as walnuts and other nuts.
- Use little or no salt in your foods. Reducing sodium (salt) can lower blood pressure and decrease the risk of hypertension, heart disease, and heart failure.
- Limit your intake of sugar. Choose nutrient-rich fruits instead of beverages and processed foods that contain added sugars.
- If you consume alcohol, do so in moderation. The American Heart Association (AHA) recommends limiting alcohol to no more than 1 drink per day for women.
Calcium and Vitamin D
A combination of calcium and vitamin D can reduce the risk of osteoporosis, the bone loss associated with menopause. The best sources are from calcium-rich and vitamin D-fortified foods.
Doctors are currently reconsidering the use of calcium and vitamin D supplements. The U.S. Preventive Services Task Force (USPSTF) advises that healthy postmenopausal women don't need to take these supplements. According to the USPSTF, taking daily low-dose amounts of vitamin D supplements (less than 400 IU), with or without calcium supplements (less than 1,000 mg), does not prevent fractures. For higher doses, the USPSTF says there is not enough evidence to make a recommendation. In addition to possible lack of benefit, these supplements are associated (even at lower doses) with certain risks, like kidney stones.
However, calcium and vitamin D are important nutrients. Supplements may be appropriate for certain people including those who do not get enough vitamin D through sunlight exposure and those who do not consume enough calcium in their diet. They are also helpful for people who have been diagnosed with osteoporosis. Talk with your doctor about whether or not you should take supplements.
The National Osteoporosis Foundation (NOF) recommends:
- Adults under age 50 should have 1,000 mg of calcium and 400 to 800 IU of vitamin D daily.
- Calcium supplements should be used only if dietary intake is not adequate. The average daily calcium intake from diet in adults over age 50 is between 600 and 700 mg per day.
- Adult men ages 50 to 70 should have 1,000 mg of calcium daily. Men over age 70 should have 1200 mg daily.
- Adult women age 50 and older should have 1,200 mg of calcium daily.
- Calcium intake above these amounts has not shown to provide additional bone strength and may increase the risk of kidney stones, heart disease, and stroke.
- 800 to 1,000 IU of vitamin D daily is recommended for adults age 50 and older. (The recommended Institute of Medicine Dietary Reference Intakes for vitamin D are 600 IU per day through age 70 and 800 IU per day over age 70.)
Calcium
Women should consume low-fat dairy products and other foods rich in calcium (dark green vegetables, sardines), or calcium-fortified foods and beverages (orange juice, cereal), to get enough calcium in their diet. Calcium supplements may be an option for women who do not consume adequate amounts of calcium in their diets. Calcium supplements include calcium carbonate (Caltrate, Os-Cal, and Tums), calcium citrate (Citracal), calcium gluconate, and calcium lactate. Although each kind provides calcium, they all have different calcium concentrations, absorption capabilities, and other actions.
High doses (over 2,500 mg per day) of calcium supplements may increase the risk for kidney stones and may possibly increase the risk for heart attack. Adults who consume adequate amounts of calcium in their diets do not need to take a calcium supplement. Taking more calcium than recommended is not helpful and may cause harmful side effects.
Vitamin D
Vitamin D is necessary for the absorption of calcium in the stomach and gastrointestinal tract and is the essential companion to calcium in maintaining strong bones.
Vitamin D is made in the skin using energy from the ultraviolet rays in sunlight. As people age, their vitamin D levels decline. Levels also fall during winter months and when people have inadequate exposure to sunlight.
Dietary sources of vitamin D include fatty fish (salmon, mackerel, tuna), egg yolks, liver, and vitamin D-fortified foods and beverages (milk, orange juice, soy milk, cereals). Older women who do not get enough vitamin D solely from diet or sunlight may need to take a supplement. Vitamin D supplements are available either as D2 (ergocalciferol) or D3 (cholecalciferol). They work equally well for bone health. Like calcium supplements, vitamin D supplements may increase the risks for kidney stones.
Calcium and vitamin D supplements can be taken as separate supplements or as a combination supplement. If separate preparations are used, they do not need to be taken at the same time. Given the controversies over the benefits and safety of these supplements, be sure to discuss with your doctor whether supplements are a good choice for you. As an alternative to supplements, many doctors recommend making dietary changes to increase calcium and vitamin D intake and getting 15 minutes a day of sun exposure.
Alcohol
Effect on the Heart
One drink a day in women who are not at risk for alcohol abuse may be beneficial for the heart. However, the American Heart Association recommends that women have no more than one drink per day.
Effect on Bones
Excessive alcohol consumption increases the risk for brittle bones and osteoporosis.
Effect on Breast Cancer
Alcohol increases the risk for breast cancer. Any woman who is at high risk should consider not drinking at all or drinking very sparingly.
Effect on Menopausal Symptoms
Alcohol can be a trigger for hot flashes.
Controlling Weight Gain
Many women need to increase physical activity and reduce caloric intake in the years before and after menopause. Weight gain is common during these years, and it can be sudden and distressing, particularly when habitual exercise and eating patterns are no longer effective in controlling weight. In addition to reducing risk factors for heart disease, weight loss may help lessen frequency and severity of hot flashes.
Exercise
Women should pursue a lifestyle that includes a balanced aerobic and weight resistance exercise program appropriate to their age and medical conditions. Brisk walking, stair climbing, hiking, dancing, weightlifting, and tai chi are all helpful. Some studies report that exercise may help reduce hot flashes. A healthy diet plus regular, consistent exercise can also help ward off the weight gain associated with menopause. Weight-bearing exercises are specifically helpful for protecting against bone loss.
Women should get at least 30 minutes of exercise each day (for weight loss, 60 to 90 minutes is preferred). While more exercise is better, any amount of exercise is helpful.
Alternative Therapies
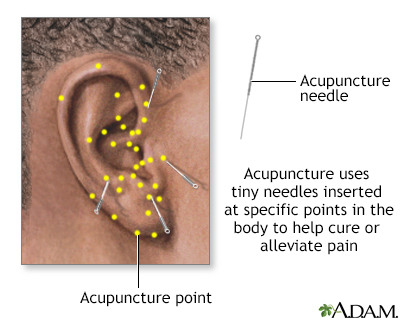
There are many unproven methods for relieving menopausal symptoms, some more effective than others. Acupuncture, meditation, yoga, and relaxation techniques are all harmless ways to reduce the stress of menopause. Some women report great benefit from these practices, but there is no scientific proof of effectiveness.
Acupuncture, hypnosis, and biofeedback are all alternative ways to control pain. Acupuncture involves the insertion of tiny sterile needles, slightly thicker than a human hair, at specific points on the body.
Herbs and Supplements
Women often try herbal or so-called natural remedies to treat menopausal symptoms. There have been numerous studies conducted on various herbal products and other complementary and alternative therapies. These studies have not found that these approaches are beneficial. Some herbs and supplements can have adverse side effects.
Phytoestrogens and Isoflavones
Many studies have researched plant estrogens (phytoestrogens), which are generally categorized as isoflavones (found in soy and red clover) and lignans (found in whole wheat and flaxseed). No evidence to date indicates that phytoestrogen foods or supplements provide benefit for hot flashes, night sweats, or other menopausal symptoms. They also do not appear to help lower cholesterol or prevent heart disease. Nevertheless, soy is a healthy food choice. The best sources of soy protein are soy food products (such as tofu, soy milk, and soybeans), not supplements.
Soy isoflavones contain genistein and daidzein, which are estrogen-like compounds. While some studies have suggested that soy food consumption may protect against estrogen-responsive cancers such as breast and endometrial cancer, other studies have indicated that high intakes of soy may increase the risks of these cancers. The American Cancer Society recommends that women with breast cancer eat only moderate amounts of soy food and avoid taking dietary supplements that contain high amounts of isoflavones.
Other Herbs and Supplements
The following herbs and dietary supplements are sometimes used for menopausal symptoms and have certain risks:
- Black cohosh (Cimicifuga racemosa), also known as squaw root, is the herbal remedy most studied for menopausal symptoms. Although it contains a plant estrogen, this substance does not act like an estrogen in the human body. Studies have shown mixed results in preventing hot flashes. High-quality studies have found that black cohosh works no better than placebo for treating hot flashes and night sweats. Headaches and gastrointestinal problems are common side effects. This herb has been associated with liver toxicity.
- Dong quai (Angelica sinensis) does not appear helpful for hot flashes or other menopausal symptoms. Do not use dong quai with blood-thinning drugs, such as warfarin, because it may cause bleeding complications.
- Ginseng (Panax ginseng) has been reported to possibly help menopausal symptoms of depression and sleep problems, but it has no effect on hot flashes.
- Kava (Piper methysticum) may relieve anxiety but it does not help hot flashes. This herb is dangerous. There have been several reports of it causing liver failure and death, especially in people with liver disease. Do not use kava.
- Wild yam (Dioscorea villosa) is an herb sometimes used for menstrual problems as well as menopausal symptoms. It contains plant progesterone. However, like black cohosh, there is no evidence that the human body can convert this substance into a hormone. Be aware that some commercial herbal wild yam products contain prescription progesterone.
- Evening primrose oil has not been shown to be superior to placebo. It may increase the risk for seizures when taken with certain types of drugs.
- Dehydroepiandrosterone (DHEA) is a weak male hormone secreted by the adrenal gland. It is available as a dietary supplement. DHEA has no benefit for hot flashes and may increase the risk of breast cancer.
Generally, manufacturers of herbal remedies and dietary supplements do not need approval from the Food and Drug Administration (FDA) to sell their products. Just like with drugs, herbs and supplements can affect the body's chemistry, and therefore have the potential to produce side effects that may be harmful. There have been a number of reported cases of serious and even lethal side effects from herbal products. Patients should check with their doctors before using any herbal remedies or dietary supplements.
- Medications
Hormone Therapy (HT)
Hormone therapy (HT), also known as menopausal hormone therapy (MHT) or hormone replacement therapy (HRT), uses medications that contain the female hormones that the body has stopped producing after menopause. The primary reasons that women use HT are for the relief of hot flashes and night sweats (vasomotor symptoms), and vaginal dryness.
Hormone therapy uses either:
- Estrogen alone (known as estrogen therapy [ET]).
- Estrogen in combination with progestogen (known as estrogen-progestogen therapy [EPT]).
Different types of estrogen are used in hormone therapy products. They include estradiol and conjugated estrogens.
The term "progestogen" encompasses both progesterone and progestin. Progesterone is the name for the natural hormone that the body produces. Progestin refers to a synthetic hormone that has progesterone effects. Because estrogen alone can increase the risk for uterine (endometrial) cancer, progestogen is added to estrogen to protect the uterine lining (endometrium) and reduce this risk.
Women receive either ET or EPT depending on whether they have a uterus:
- Women who have a uterus (have not had a hysterectomy) receive estrogen plus progesterone or a progestin (EPT).
- Women who do not have a uterus (have had a hysterectomy) receive estrogen alone (ET).
General Recommendations for HT
Current guidelines support the use of HT for the treatment of severe hot flashes that do not respond to non-hormonal therapies. General recommendations include:
- HT may be started in women who have recently entered menopause.
- HT should not be used in women who have started menopause many years ago.
- Women should not take HT (either EPT or ET) if they have risks for stroke, heart disease, blood clots, and breast cancer.
- Currently, there is no consensus on how long HT should be used or at what age it should be discontinued. Treatment should be individualized for a woman's specific health profile.
- HT should be used only for menopause symptom management, not for chronic disease prevention.
Initiating Therapy
Before starting HT, your doctor should give you a comprehensive physical exam and take your medical history to evaluate your risks for:
- Heart disease
- Stroke
- Blood clots
- Osteoporosis
- Breast cancer
While taking HT, you should have regular mammograms and pelvic exams and Pap smears. Current guidelines recommend that if HT is needed, it should be initiated around the time of menopause. Studies indicate that the risk of serious side effects is lower for women who use HT while in their 50s. Women who start HT past the age of 60 appear to have a higher risk for side effects such as heart attack, stroke, blood clots, or breast cancer. HT should be used with care in this age group.
Women who experience premature menopause are usually prescribed HT or oral contraceptives to help prevent bone loss. These women should be reevaluated when they reach the age of natural menopause (around age 51) to determine whether they should continue to take hormones.
Discontinuing Therapy
When a woman stops taking HT, perimenopausal symptoms may recur. There is about a 50% chance of hot flashes recurring regardless of whether HT is suddenly stopped or gradually tapered off. When a woman reaches full menopause, symptoms will eventually go away.
Because HT offers protection against osteoporosis, when women stop taking HT their risks for bone thinning and fractures increases. For women who have used HT for several years, doctors should monitor their bone mineral density and prescribe bone-preserving medications if necessary.
Safety Concerns
Until 2002, doctors used to routinely prescribe HT to reduce the risk of heart disease and other health risks in addition to treating menopausal symptoms. That year, the results of an important study, called the Women's Health Initiative (WHI), led doctors to revise their recommendations regarding HT.
The WHI, started in 1991, is an on-going health study of nearly 162,000 postmenopausal women. Part of the study focuses on the benefits and risks of hormone therapy. As new data are released and analyzed, there have been a number of changes in the way HT is prescribed and a better understanding of its risks.
Women who should not take hormone therapy include those with the following conditions:
- Current, past, or suspected breast cancer
- History of endometrial cancer
- Vaginal bleeding of unknown cause
- Current or past history of blood clots
- High blood pressure that is untreated or poorly managed
- History of angina, heart attack, or other heart or circulation problems
HT Forms and Regimens
HT comes in several forms:
- Oral tablets or pills
- Skin patches
- Vaginal cream or tablet
- Vaginal ring
- Topical gel or spray
HT pills and skin patches are considered "systemic" therapy because the medication delivered affects the entire body. The risk for blood clots, heart attacks, and certain types of cancers is higher with hormone pills than with skin patches or other transdermal forms.
Vaginal forms of HT are called "local" therapy. Doctors generally prescribe vaginal applications of low-dose estrogen therapy to specifically treat menopausal symptoms such as vaginal dryness and pain during sex. This type of ET is available in a cream, tablet, or ring that is inserted into the vagina.
"Bioidentical" Hormones
"Bioidentical" hormone therapy is promoted as a supposedly more natural and safer alternative to commercial prescription hormones. Bioidentical hormones are typically compounded in a pharmacy. Some compounding pharmacies claim that they can customize these formulations based on saliva tests that show a woman's individual hormone levels.
The FDA and many professional medical associations warn patients that "bioidentical" is a marketing term that has no scientific validity. Formulations sold in these pharmacies have not undergone FDA regulatory scrutiny. Some of these compounds contain estriol, a weak form of estrogen, which has not been approved by the FDA for use in any drug. In addition, saliva tests do not give accurate or realistic results, as a woman's hormone levels fluctuate throughout the day.
FDA-approved hormones available by prescription come from different synthetic and natural sources, including plant-based. (For example, Prometrium is a progesterone derived from yam plants.)
Benefits of HT
Perimenopausal and Menopausal Symptoms
Systemic HT is mainly recommended for relieving menopausal symptoms such as hot flashes, night sweats, and sleep problems, as well as vaginal dryness. Local HT (delivered vaginally) is used specifically for treating vaginal dryness and atrophy; and accompanying pain during sexual intercourse. HT does not prevent certain other problems associated with menopausal changes, such as thinning hair or weight gain. It is unclear whether HT helps improve mood.
Osteoporosis
Estrogen increases and helps maintain bone density. HT may be useful for some women at high risk for osteoporosis, but for most women the risks do not outweigh the benefits. Other drugs, such as bisphosphonates, should be considered first-line treatment for osteoporosis. Duavee is a drug that contains a combination of conjugated equine estrogen and the selective estrogen receptor modulator (SERM) bazedoxifene. It is approved to treat hot flashes and prevent osteoporosis in women with a uterus.
Although HT may have some benefits in addition to menopausal symptoms, results from the Women's Health Initiative (WHI) studies strongly indicate that HT should be used only for relief of menopausal symptoms, not for prevention of chronic disease.
Risks of HRT
Heart Disease, Heart Attack, and Stroke
HT may increase the risk of heart disease and heart attack in older women, or in women who began estrogen use more than 10 years after their last period. HT is probably safest in healthy women under age 60 who begin taking it within 10 years after the start of menopause. Taking HT in order to prevent heart disease is not recommended. Women who have a history of heart disease or heart attack should not take HT. HT may increase the risk of stroke.
Blood Clots
HT increases the risk for formation of blood clots in the veins (deep venous thrombosis) or in the lungs (pulmonary embolism). The risk for blood clots is higher with oral forms of HT than with transdermal forms (skin patches, creams). There appears to be little, if any, increase in the risk of blood clots when transdermal forms of HT are used.
Breast Cancer
Estrogen- progestogen therapy (EPT) increases the risk for breast cancer if used for more than 3 to 5 years. This risk appears to decline within 3 years of stopping combination HT.
Estrogen-only therapy (ET) does not significantly increase the risk of developing breast cancer if it is used for less than 7 years. If used for more than 7 years, it may increase the risk of breast (and ovarian) cancers, especially for women already at increased risk for breast cancer. The North American Menopause Society does not recommend ET use in breast cancer survivors as it has not been proven safe and may raise the risk of recurrence.
Both estrogen-only and combination HT increase breast cancer density, making mammograms more difficult to read. This can cause cancer to be diagnosed at a later stage. Women who take HT should be aware of the need for regular mammogram screenings.
The North American Menopause Society recommends that women who are at risk for breast cancer avoid hormone therapy and try other options to manage menopausal symptoms.
Ovarian Cancer
Long-term use (more than 5 to 10 years) of estrogen-only therapy (ET) may increase the risk of developing and dying from ovarian cancer. The risk is less clear for combination estrogen-progesterone therapy (EPT).
Endometrial (Uterine) Cancers
Taking estrogen-only therapy (ET) for more than 3 years significantly increases the risk of endometrial cancer. If taken for 10 years, the risk is even greater. Adding progesterone to estrogen (EPT) helps to reduce this risk. Women who take ET should anticipate uterine bleeding, especially if they are obese, and may need endometrial biopsies and other gynecologic tests. No type of hormone therapy is recommended for women with a history of endometrial cancer.
Lung Cancer
It is not clear if HT use is associated with an increased risk of lung cancer, women who smoke and who are past or current users of HT should be aware that that EPT may possibly promote the growth of lung cancers.
Gallbladder Disease
HT increases the risk of developing gallbladder disease.
Cognitive Function
The Women's Health Initiative Memory Study and other studies suggest that combined HT does not reduce the risk of cognitive impairment or dementia and may actually increase the risk of cognitive decline. Researchers are continuing to study the effects of HT on Alzheimer disease risk.
Other Drugs Used for Menopausal Symptoms
Despite its risks, hormone therapy appears to be the most effective treatment for hot flashes. There are, however, nonhormonal treatments for hot flashes and other menopausal symptoms.
Antidepressants
The antidepressants known as selective serotonin-reuptake inhibitors (SSRIs) are sometimes used for managing mood changes and hot flashes. A low-dose formulation of paroxetine (Brisdelle) is approved to treat moderate-to-severe hot flashes associated with menopause. Other SSRIs and similar antidepressant medicines are used "off-label" and may have some benefit too. They include fluoxetine (Prozac, generic), sertraline (Zoloft, generic), venlafaxine (Effexor), desvenlafaxine (Pristiq), paroxetine (Paxil, generic), and escitalopram (Lexapro, generic).
Gabapentin
Several small studies have suggested that gabapentin (Neurontin), a drug used for seizures and nerve pain, may relieve hot flashes. This drug is sometimes prescribed "off-label" for treating hot flash symptoms. However, in 2013 the FDA decided against approving gabapentin for this indication because the drug demonstrated only modest benefit. Gabapentin may cause:
- Drowsiness
- Dizziness
- Fatigue
- Swelling of the hands and feet
Clonidine
Clonidine (Catapres, generic) is a drug used to treat high blood pressure. Studies show it may help manage hot flashes. Side effects include dizziness, drowsiness, dry mouth, and constipation.
Testosterone
Some doctors prescribe combinations of estrogen and small amounts of the male hormone testosterone to improve sexual function and increase bone density. Side effects of testosterone therapy include:
- Increased body hair
- Acne
- Fluid retention
- Anxiety
- Depression
Testosterone also adversely affects cholesterol and lipid levels, and combined estrogen and testosterone may increase the risk of breast cancer. Many experts do not consider testosterone safe or effective for treatment of menopausal symptoms.
Non-Hormonal Treatments for Vaginal Dryness and Atrophy
Vaginal lubricants (such as KY Jelly and Astroglide) and moisturizers (such as Replens) can be purchased without a prescription and are safe and helpful for treating vaginal dryness and dyspareunia (painful sexual intercourse). Dyspareunia is a result of thinning vaginal tissues (vaginal atrophy) due to low estrogen levels.
The North American Menopause Society recommends lubricants and long-acting moisturizers as first-line treatments for vaginal atrophy. For women who still experience discomfort, low-dose vaginal (local) estrogen is the next option.
Ospemifene (Osphena) is approved as a non-hormonal prescription drug for treating menopausal-associated vaginal dryness and dyspareunia. Ospemifene is an oral drug (pill) that acts like an estrogen on vaginal tissues to make them thicker and less fragile. However, this drug may cause the lining of the uterus (endometrium) to thicken, which can increase the risk for uterine (endometrial) cancer. Because of this and other risks, ospemifene should only be taken for a short amount of time. Common side effects of ospemifene include hot flashes, vaginal discharge, and excessive sweating.
- Resources
- North American Menopause Society -- www.menopause.org
- American College of Obstetricians and Gynecologists -- www.acog.org
- National Institute on Aging -- www.nia.nih.gov
- National Institutes of Health - Menopausal Hormone Therapy Information -- www.nih.gov/health-information/menopausal-hormone-therapy-information
References
American College of Obstetricians and Gynecologists. ACOG committee opinion no. 556: postmenopausal estrogen therapy: route of administration and risk of venous thromboembolism. Obstet Gynecol. 2013;121(4):887-890. PMID: 23635705 www.ncbi.nlm.nih.gov/pubmed/23635705.
American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 141: management of menopausal symptoms. Obstet Gynecol. 2014;123(1):202-216. PMID: 24463691 www.ncbi.nlm.nih.gov/pubmed/24463691.
Committee on Gynecologic Practice and the American Society for Reproductive Medicine Practice Committee. Committee opinion no. 532: compounded bioidentical menopausal hormone therapy. Obstet Gynecol. 2012;120(2 Pt 1):411-415. PMID: 22825109 www.ncbi.nlm.nih.gov/pubmed/22825109.
Dodin S, Blanchet C, Marc I, et al. Acupuncture for menopausal hot flushes. Cochrane Database Syst Rev. 2013;7:CD007410. PMID: 23897589 www.ncbi.nlm.nih.gov/pubmed/23897589.
Hill DA, Crider M, Hill SR. Hormone therapy and other treatments for symptoms of menopause. Am Fam Physician. 2016;94(11):884-889. PMID: 27929271 www.ncbi.nlm.nih.gov/pubmed/27929271.
Hodis HN, Mack WJ, Henderson VW, et al. Vascular effects of early versus late postmenopausal treatment with estradiol. N Engl J Med. 2016;374(13):1221-1231. PMID: 27028912 www.ncbi.nlm.nih.gov/pubmed/27028912.
Lamberts SWJ, van den Beld AW. Endocrinology and aging. In: Melmed S, Polonsky KS, Larsen PR, Kronenberg HM, eds. Williams Textbook of Endocrinology. 13th ed. Philadelphia, PA: Elsevier; 2016:chap 27.
Lethaby A, Marjoribanks J, Kronenberg F, Roberts H, Eden J, Brown J. Phytoestrogens for menopausal vasomotor symptoms. Cochrane Database Syst Rev. 2013;12:CD001395 PMID: 24323914 www.ncbi.nlm.nih.gov/pubmed/24323914.
Lobo RA. Menopause and care of the mature woman: endocrinology, consequences of estrogen deficiency, effects of hormone therapy, and other treatment options. In: Lobo RA, Gershenson DM, Lentz GM, Valea FA, eds. Comprehensive Gynecology. 7th ed. Philadelphia, PA: Elsevier; 2017:chap 14.
Lobo RA. Hormone-replacement therapy: current thinking. Nat Rev Endocrinol. 2017;13(4):220-231. PMID: 27716751 www.ncbi.nlm.nih.gov/pubmed/27716751.
Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women's Health Initiative randomized trials. JAMA. 2013;310(13):1353-1368. PMID: 24084921 www.ncbi.nlm.nih.gov/pubmed/24084921.
Moyer VA; US Preventive Services Task Force. Menopausal hormone therapy for the primary prevention of chronic conditions: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2013;158(1):47-54. PMID: 23090711 www.ncbi.nlm.nih.gov/pubmed/23090711.
Moyer VA; US Preventive Services Task Force. Vitamin D and calcium supplementation to prevent fractures in adults: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2013;158(9):691-696. PMID: 23440163 www.ncbi.nlm.nih.gov/pubmed/23440163.
Management of symptomatic vulvovaginal atrophy: 2013 position statement of The North American Menopause Society. Menopause. 2013;20(9):888-902. PMID: 23985562 www.ncbi.nlm.nih.gov/pubmed/23985562.
NAMS 2017 Hormone Therapy Position Statement Advisory Panel. The 2017 hormone therapy position statement of The North American Menopause Society. Menopause. 2017;24(7):728-753. PMID: 28650869 www.ncbi.nlm.nih.gov/pubmed/28650869.
Santoro N, Neal-Perry G. Menopause. In: Goldman L, Schafer AI, eds. Goldman-Cecil Medicine. 26th ed. Philadelphia, PA: Elsevier; 2020:chap 240.
Skaznik-Wikiel ME, Traub ML, Santoro N. Menopause. In: Jameson JL, De Groot LJ, de Kretser DM, et al, eds. Endocrinology: Adult and Pediatric. 7th ed. Philadelphia, PA: Elsevier Saunders; 2016:chap 135.